First Quarter’s Highlights (Year Over Year):
- Total revenue in the first fiscal quarter increased to $27.7 million, or 5%, from $26.3 million in the prior year period.
- Clinical Labs revenue totaled $20.3 million, or an increase of 10% from $18.6 million a year ago.
- Gross profit increased 1%, with operating income improving $0.7 million, or 53%. Gross margin was 44%.
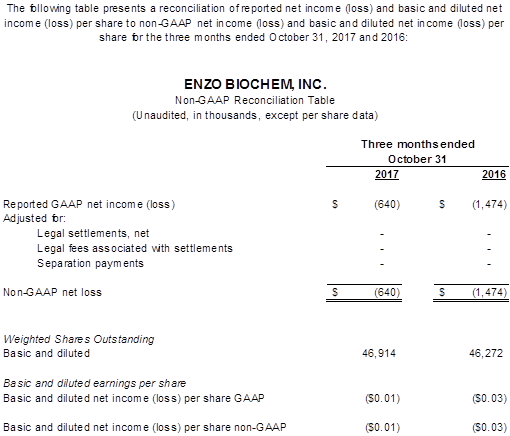
- The GAAP and non-GAAP net loss was $0.6 million or $(0.01) per fully diluted share, an improvement of $0.8 million, or 56% over the prior year’s net loss of $1.5 million or $(0.03) per fully diluted share.
- Total cash and cash equivalents at October 31, 2017 was $66.8 million, an increase of $2.7 million from July 31, 2017. Tight cost controls and efficiencies resulted in positive $2.6 million cash flow from operations in the quarter.
- During the quarter, the New York State Health Department issued conditional approval of the final three women’s health related molecular diagnostic tests which completes a 13-analyte panel, placing Enzo in a leading position to benefit from higher-margin proprietary women’s health diagnostics.
- Plant facilities in Farmingdale, NY, undergoing expansion to accommodate anticipated growth in manufacturing of growing roster of new molecular diagnostic assays and to increase laboratory capacity and to support GMP certification.
Barry Weiner, President, Comments:
“The first quarter of fiscal 2018 saw additional progress in moving aggressively towards our model as an integrated, growth-oriented molecular diagnostics company, and a low-cost medically related assay provider and reference service organization. Revenues continued to grow, spurred by double digit growth in the quarter at Clinical Labs, the result of expanded product and service capabilities and geographical expansion, along with increasing volume from our association with leading healthcare insurance providers. Sales, general and administrative costs (SG&A) are being held in check, even as product development and research efforts continue. We are also completing preparations to launch our marketing and sales program of our comprehensive products and reference services menu, while also adding to our Lab’s capability, particularly in the women’s health diagnostic field.
“We are preparing to offer one of the industry’s most advanced and well-rounded 13-analyte women’s health diagnostics panel, which last month was presented at the annual meeting of the prestigious Association for Molecular Pathology. The study our group reported on demonstrated that Enzo’s AMPIPROBE® technology provides a new proprietary approach to creating clinically sensitive and specific multiplex real-time PCR assays, which, when run in tandem, can serve as a comprehensive panel for the accurate detection and identification of vaginitis-associated microorganisms. Enhanced by our proprietary AMPIPROBE® technology, it offers faster, more specific and more sensitive molecular vaginitis testing, which we expect to improve diagnoses of vaginitis, a leading medical concern among women. Increasingly, when measured against the performance of similar assays, Enzo products continue to outperform, as reported recently in a leading diagnostic pathology publication, which cited our Polyview™ technology for viewing the clinical morphology of HPV specimens as having no false positives. That compared favorably to other leading competitive products that did have false positives in their tests.
“Our focus is on gaining sustained profitability and advancing healthcare in today’s challenging environment by providing affordable and reliable diagnostic testing. With Medicare reimbursement rates expected to decline materially over the next three years based on a new formula being adopted in place of one used over the past 30 years, the challenges are clear. Enzo’s transformative blueprint may help to alleviate this new challenge to the nation’s independent labs. This opportunity is reflected in our expanding line of medically related, versatile, highly efficient platforms and assays that are increasingly professionally recognized for their sensitivity and economics.”
First Quarter Results
Total revenues increased to $27.7 million or 5% higher than the prior year period driven by increased Clinical labs services revenue from MDx tests. Gross profit was $12.2 million or 44% of total revenue. Total operating expenses were $12.9 million, a decline of $0.5 million or 4% over a year ago.
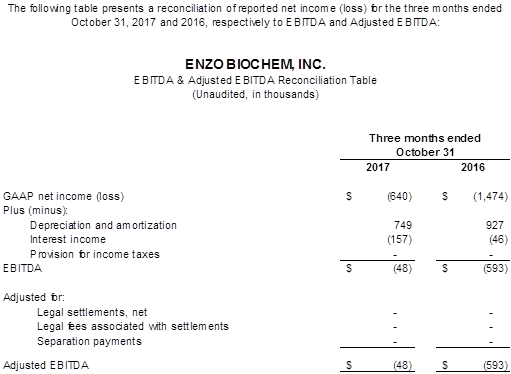
The GAAP and Non-GAAP net loss amounted to $(0.6) million, or $(0.01) per fully diluted share, compared to the year ago net loss of $(1.5) million or $(0.03) per fully diluted share, an improvement of $0.8 million. EBITDA and Adjusted EBITDA were essentially break even compared with a loss of $(0.6) million a year ago, a $0.5 million improvement year over year.
As of October 31, 2017 working capital amounted to $71.4 million, cash and cash equivalents totaled $66.8 million, up $2.7 million from three months earlier. Apart from capital leases, there was no debt.
Quarterly Segment Results
Enzo Clinical Labs revenues increased to $20.3 million, or 10%, from $18.6 million a year ago. As noted above, expansion of its diagnostics services to additional nearby geographical locations, the increased product mix of diagnostic tests, particularly in the women’s health field, and greater volume, all are contributing to its steady test and service growth. Gross profit increased to $8.3 million, from $7.7 million, with gross margin as a percentage of revenues at 41% for both the current and year ago quarters. Total operating expenses increased by 3.6%, to $6.9 million. Operating income amounted to $1.4 million, a nearly 40% increase.
Enzo Life Sciences revenues were $7.1 million compared to $7.4 million a year ago, a decline of $0.3 million or 5%. Gross profit approximated $4 million and gross margin as a percentage of revenue was 54%, compared to $4.4 million and 57%, respectively, a year ago. With the continued focus on reducing costs and improving efficiency, SG&A declined by 11%, to $2.6 million, with total operating expenses declining 12%, to $3.2 million.
Conference Call
The Company will conduct a conference call Friday, December 8, 2017 at 8:30 AM ET. The call can be accessed by dialing 1-888-459-5609. International callers can dial 1-973-321-1024. Please reference PIN number 3678737.
Interested parties may also listen over the Internet at: https://tinyurl.com/yd96bbmv
To listen to the live call on the Internet, please go to the web site at least fifteen minutes early to register, download and install any necessary audio software. For those who cannot listen to the live broadcast, a replay will be available approximately two hours after the end of the live call, through midnight (ET) on December 22, 2017. The replay of the conference call can be accessed by dialing 1-855-859-2056, and when prompted, use PIN number 3678737. International callers can dial 1-404-537-3406, using the same PIN number.
NON-GAAP Financial Measures
To comply with Regulation G promulgated pursuant to the Sarbanes-Oxley Act, Enzo Biochem attached to this news release and will post to the Company's investor relations web site (www.enzo.com) any reconciliation of differences between non-GAAP financial information that may be required in connection with issuing the Company's quarterly financial results.
The Company uses EBITDA as a measure of performance to demonstrate earnings exclusive of interest, taxes, depreciation and amortization. Adjustments to EBITDA are for items of a non-recurring nature and are reconciled on the table provided. The Company manages its business based on its operating cash flows. The Company, in its daily management of its business affairs and analysis of its monthly, quarterly and annual performance, makes its decisions based on cash flows, not on the amortization of assets obtained through historical activities. The Company, in managing its current and future affairs, cannot affect the amortization of the intangible assets to any material degree, and therefore uses EBITDA as its primary management guide. Since an outside investor may base its evaluation of the Company's performance based on the Company's net loss not its cash flows, there is a limitation to the EBITDA measurement. EBITDA is not, and should not be considered, an alternative to net loss, loss from operations, or any other measure for determining operating performance of liquidity, as determined under accounting principles generally accepted in the United States (GAAP). The most directly comparable GAAP reference in the Company's case is the removal of interest, taxes, depreciation and amortization.
We refer you to the tables attached to this press release which includes reconciliation tables of GAAP to Non-GAAP net income (loss) and EBITDA to Adjusted EBITDA.
About Enzo Biochem
Enzo Biochem is a pioneer in molecular diagnostics, leading the convergence of clinical laboratories, life sciences and intellectual property through the development of unique diagnostic platform technologies that provide numerous advantages over previous standards. A global company, Enzo Biochem utilizes cross-functional teams to develop and deploy products, systems and services that meet the ever-changing and rapidly growing needs of health care today and into the future. Underpinning Enzo Biochem’s products and technologies is a broad and deep intellectual property portfolio, with patent coverage across a number of key enabling technologies.
Except for historical information, the matters discussed in this news release may be considered "forward-looking" statements within the meaning of Section 27A of the Securities Act of 1933, as amended and Section 21E of the Securities Exchange Act of 1934, as amended. Such statements include declarations regarding the intent, belief or current expectations of the Company and its management, including those related to cash flow, gross margins, revenues, and expenses which are dependent on a number of factors outside of the control of the Company including, inter alia, the markets for the Company’s products and services, costs of goods and services, other expenses, government regulations, litigation, and general business conditions. See Risk Factors in the Company’s Form 10-K for the fiscal year ended July 31, 2017. Investors are cautioned that any such forward-looking statements are not guarantees of future performance and involve a number of risks and uncertainties that could materially affect actual results. The Company disclaims any obligations to update any forward-looking statement as a result of developments occurring after the date of this press release.
###
Contact:
For: Enzo Biochem, Inc.
Steve Anreder, 212-532-3232 or Michael Wachs, CEOcast, Inc., 212-732-4300
steven.anreder@anreder.com mwachs@ceocast.com