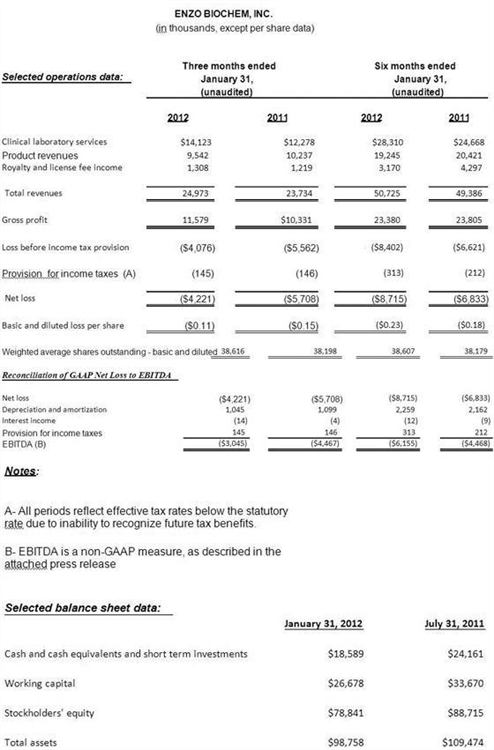
- Total revenues increased 5% year over year, led by strong organic growth at Enzo Clinical Labs and increased royalty and licensee income;
- Gross margin was up 12%;
- Operating results improved by $1.5 million;
- Approval received by New York State Department of Health for ColonSentry, a proprietary assay for assessment of a patient’s risk of having colorectal cancer;
- AmpiProbe™ HCV Assay, first of Company’s proprietary nucleic acid amplification technology, to be unveiled at leading European medical meeting.
“The quarter was another period of progress, both on the operational and development fronts. Enzo Clinical Labs posted another quarter of strong organic growth. At Enzo Life Sciences margins have begun improving as our efforts to streamline the product mix, improve marketing and enhance manufacturing start to yield results. Our recent announcements related to molecular diagnostics further support our progress in the development of novel assays that hold promise to contribute to the advancement of healthcare,” said Barry Weiner, President.
Second Quarter Results
Revenues for the second fiscal quarter were $25.0 million, compared with $23.7 million a year ago, an increase of 5%, as a result of both continued growth at Enzo Clinical Labs and an increase in royalty and licensing fee income. Gross profit improved to 46%, from 44% year over year. Operating expenses declined $0.5 million to $15.4 million, due to a reduction in legal and R&D expenses, partially offset by an increased provision for uncollectible accounts receivable due to higher service volume at Enzo Clinical Labs. However, the Company increased its developmental effort in the area of new molecular tests and technology platform development. Selling, general and administrative expenses as a percentage of revenues declined to 46% from 49% in the year ago period. The net loss for the period was reduced by $1.5 million or ($.04) per diluted share, declining to $4.2 million or ($.11) loss per diluted share. EBITDA loss was lower by $1.4 million or 32% to $3.0 million. Total cash used was $2.8 million, which included a non-recurring cash payment of $1.1 million for the final earn-out from a previous acquisition and cash used in operations of $1.3 million.
As of January 31, 2012, cash, cash equivalents and short term investments totaled $18.6 million. Working capital amounted to $26.7 million. There was no long-term debt.
Segment Quarterly Results
Enzo Clinical Labs increased revenues organically by $1.8 million or 15%, as the Lab continued to expand marketing, reaching more physicians and increasing specimen volume. Gross margin was up 13%, to $5.4 million, however higher costs impacted the gross profit percentage by 100 basis points. Increases in SG&A from staff additions and higher commission levels were both driven from the higher service volume. The increase in provision for uncollectible accounts receivables was due to greater patient service volume, although as a percentage of revenue the provision was comparable in both periods at 8%.
At Enzo Life Sciences, operating income rose, as a result of a reduction in expenses and improved operations. Revenues amounted to $9.5 million as compared to $10.2 million, due largely to the continued softness in the academic market and certain distributed products. The Company continues to achieve benefits from its program to integrate the previously acquired global businesses, and focus on greater efficiencies in manufacturing, which drove improved gross profit as a percentage of revenues. Royalty and license fee income improved 7% to $1.3 million. Operating income improved by $1.1 million to $0.7 million, compared to a year ago operating loss of $0.4 million.
Year-to –Date Highlights
Year to date revenues advanced by $1.3 million or 3% to $50.7 million.
Enzo Clinical Labs’ revenues experienced 15% organic growth while Enzo Life Sciences’ product revenues declined 6% and royalty and licensing revenues declined $1.1 million, largely due to previously reported reduced first quarter royalty and license fees. Gross margins were $23.4 million, 2% lower than a year ago also impacted by royalty and licensing fees. Operating expenses, including R&D, SG&A, legal and provision for uncollectible accounts, increased by 4%, with higher selling costs and provision for uncollectible accounts receivables attributed to increased service volume at Clinical Labs. The year-to-date loss amounted to $8.7 million, compared to $6.8 million in the corresponding year-ago period. Cash used in operations was $3.6 million.
Other Developments
As reported previously, the New York State Department of Health has approved Enzo Clinical Labs’ use of the ColonSentry™ test for providing an assessment of a patient’s risk of having colorectal cancer. The information provided by this test can guide physicians’ recommendations to patients for a colonoscopy, which remains highly underutilized by many individuals.
On March 31, 2012, Enzo will make its first peer-reviewed presentation of its novel AmpiProbe™ technology platform at the annual meeting in London of the prestigious European Society of Clinical Microbiology and Infectious Diseases (ESCMID). The platform can provide for the development of low cost high sensitivity, real time nucleic acid amplification assays. Initially, the plan is to seek CE-IVD (Confromité Européenne in vitro diagnostic) designation for AmpiProbe™ HCV the first assay based on this platform, which would allow marketing of this product as a diagnostic throughout the European Union. Application for approval by the New York State Department of Health as a laboratory developed test is also planned.
Conference Call
The Company will conduct a conference call on March 13, 2012 at 8:30 AM ET. The call can be accessed by dialing 1-888-459-5609. International callers can dial 1-973-321-1024. Please reference PIN number 59249560. Interested parties may also listen over the Internet at http://phoenix.corporate-ir.net/phoenix.zhtml?p=irol-eventDetails&c=94391&eventID=4734348. To listen to the live call on the Internet, please go to the web site at least fifteen minutes early to register, download and install any necessary audio software. For those who cannot listen to the live broadcast, a replay will be available approximately two hours after the end of the live call, through midnight (ET) on March 27, 2012. The replay of the conference call can be accessed by dialing 1-800-585-8367, and when prompted, use PIN number 59249560. International callers can dial 1-404-537-3406, using the same PIN number.
NON-GAAP Financial Measures
To comply with Regulation G promulgated pursuant to the Sarbanes-Oxley Act, Enzo Biochem attached to this news release and will post to the Company's investor relations web site (www.enzo.com) any reconciliation of differences between non-GAAP financial information that may be required in connection with issuing the Company's quarterly financial results.
The Company uses EBITDA, as a measure of performance to demonstrate earnings exclusive of interest, taxes, depreciation and amortization. The Company manages its business based on its operating cash flows. The Company, in its daily management of its business affairs and analysis of its monthly, quarterly and annual performance, makes its decisions based on cash flows, not on the amortization of assets obtained through historical activities. The Company, in managing its current and future affairs, cannot affect the amortization of the intangible assets to any material degree, and therefore uses EBITDA as its primary management guide. Since an outside investor may base its evaluation of the Company's performance based on the Company's net loss not its cash flows, there is a limitation to the EBITDA measurement. EBITDA is not, and should not be considered, an alternative to net loss, loss from operations, or any other measure for determining operating performance of liquidity, as determined under accounting principles generally accepted in the United States (GAAP). The most directly comparable GAAP reference in the Company's case is the removal of interest, taxes, depreciation and amortization.
About Enzo Biochem
Enzo Biochem is a pioneer in molecular diagnostics, leading the convergence of clinical laboratories, life sciences and therapeutics through the development of unique diagnostic platform technologies that provide numerous advantages over previous standards. A global company, Enzo Biochem utilizes cross-functional teams to develop and rapidly deploy products systems and services that meet the ever-changing and rapidly growing needs of health care both today and into the future. Underpinning Enzo Biochem’s products and technologies is a broad and deep intellectual property portfolio, with patent coverage across a number of key enabling technologies.
Except for historical information, the matters discussed in this news release may be considered "forward-looking" statements within the meaning of Section 27A of the Securities Act of 1933, as amended and Section 21E of the Securities Exchange Act of 1934, as amended. Such statements include declarations regarding the intent, belief or current expectations of the Company and its management, including those related to cash flow, gross margins, revenues, and expenses are dependent on a number of factors outside of the control of the company including, inter alia, the markets for the Company’s products and services, costs of goods and services, other expenses, government regulations, litigations, and general business conditions. See Risk Factors in the Company’s Form 10-K for the fiscal year ended July 31, 2011. Investors are cautioned that any such forward-looking statements are not guarantees of future performance and involve a number of risks and uncertainties that could materially affect actual results. The Company disclaims any obligations to update any forward-looking statement as a result of developments occurring after the date of this press release.

###
Contact:
For Enzo Biochem, Inc.
Steven Anreder
Anreder & Company
212-532-3232
steven.anreder@anreder.com
or
Michael Wachs
CEOcast, Inc.
212-732-4300
mwachs@ceocast.com