Molecular Assay Approvals from New York State Department of Health and Planned Acquisition of New Facility Highlight Progress Advancing Growth Strategy
NEW YORK, NY, October 15, 2018 – Enzo Biochem Inc. (NYSE:ENZ), an integrated diagnostic and therapeutics company, today reported results for the fourth quarter and fiscal year ended July 31, 2018, in addition to announcing New York State department of Health’s approval of additional assays for new women’s health infectious disease panel continuing to drive focus in development of lower cost products, platforms and services for the clinical laboratory market.
Recent Developments
- Progress in molecular amplification and immunohistochemistry platforms is leading to full system solution to aid in addressing challenge of maintaining profitability for clinical labs in a market affected by declining reimbursement and high operating costs.
- New York State Department of Health has approved an additional three women’s health infectious disease diagnostic assays to expand Enzo’s women’s health panel to 16 pathogens on the Company’s proprietary, versatile and cost effective AMPIPROBE® platform. This addition makes the panel one of the most comprehensive in the $800 million market. This is performed using a single swab and now includes Ureaplasma spp./M. genitalium/M. hominis (UMM) in addition to Chlamydia trachomatis, Neisseria gonorrhoeae, Trichomonas vaginalis Candida spp (C. albicans, C. glabrata, C. krusei, C. parapsilosis, C. tropicalis), Atopobium vaginae, Gardnerella vaginalis, Lactobacillus spp, Megasphera spp and BVAB2. UMM testing is vital to women’s health as mycoplasmas are a significant cause of non-gonococcal urethritis. Ureaplasmas are also associated with urethritis, and a myriad of additional medical conditions.
- Entered into an agreement to purchase an additional commercial facility with nearly 36,000 square feet in Farmingdale, NY. The building adjacent to the Company’s current Long Island campus enhances the infrastructure needed to produce and distribute Enzo’s expanding low cost, open architecture diagnostic platform products and broaden related services. The Company’s platform development includes automation-compatible reagent systems and associated products for sample collection and processing through to analysis.
- Continued product and platform development activities directed to each step of the clinical testing process, expanding into sample collection and processing. The Company’s programs include manufacturing all components required for each step in the diagnostic process for integration into an open platform. Enzo’s system solutions will enable clinical laboratories to gain economic return in the diagnostics market where declining reimbursements and rigid costs from suppliers currently prevail.
- Building on prior progress in the molecular diagnostics and immunohistochemistry areas, Enzo also recently announced the validation of three clinically relevant, cost-efficient immunohistochemistry (IHC) biomarker detection tests for charting the progression of various cancers, especially in the field of women’s health. These tests operate with the Company’s open system workflow and complement Enzo’s strategy of introducing lower cost testing solutions to the global IHC market that is projected to reach over $2 billion by 2021.
- The Company recently designated an in-network laboratory provider for three new health insurance providers in the MidAtlantic and New England areas to support geographical expansion. These additional contracts are expected to add to Enzo’s national reach of testing services to millions of covered lives across the U.S.
- New York Federal case against Hoffman LaRoche proceeding with trial date set for early April 2019.
- Validation through clinical trails’ of Enzo’s fully automated, high-throughput instrumentation including sample collection and sample processing and reagents systems both for New York State and FDA.
- Completion of buildout of GMP manufacturing capabilities.
- Approval of additional assays to expand Enzo’s test menu.
- Expansion of sales and marketing, logistics and IT efforts to grow national reference laboratory accounts.
- Partnerships and collaborations with potential strategic and institutional partners to enhance commercialization and market penetration of Enzo’s high technology platforms and products.
“Fiscal 2018 was a year of solid progress in our strategic plan, and one in which each of the principal operating units posted volume increases and achieved objectives towards our growth initiatives. These initiatives began over three years ago now provide the potential for multiple ventures and partnerships that could generate significant shareholder value. Our focus is centered on providing cost efficient products and services utilizing our proprietary assays optimized for our automated, open system platforms that are compatible with existing sample collection devices as well as our own lower cost option. These have been designed to provide not only high-performance and adaptable solutions to existing lab workflow, but also to address a critical need for lower cost solutions. Besides the number of assays already approved, in process is the development of a screening assay for oncogenic forms of HPV, among others. Today’s clinical laboratories including Enzo face a two-fold challenge - to maintain revenue growth and profitability, amidst declining reimbursements and unreasonably high operating costs. The latter stem from “closed system” diagnostics platforms, sample collection devices and accessories that are a barrier to the use of low-cost and efficient third-party solutions.
Our efforts were not limited to laboratory product development, but also included building an internal sales force that will enable us to grow this business. We also have added to our development staff, and hired experienced professionals especially knowledgeable about health care providers in order to bring them into our fold as clients. In the next few quarters, our strategic plans call for a number of new tests currently under development to be submitted for New York State Department of Health for conditional approval to add to those already authorized, which will further expand our already sizable test menu. Our Women’s Health Panel was expanded with three more tests this quarter to 16 analytes that can be processed in one sample.
Our acquisition of a new facility ties in directly with the anticipated expansion of our capabilities at the Farmingdale, NY campus. In addition to enhancing efficiency, the purchase represents an important vote of confidence in our strategic plan. The new facility will provide Good Manufacturing Practices (GMP) and ISO compliant manufacturing and logistics space for Enzo’s diagnostics and life science products business. Moreover, the new facility will enhance space for GMP production of the Company’s development-stage clinical candidates, including Enzo’s proprietary sphingosine kinase 1 inhibitor, SK1-I, which is being investigated for potential applications in oncology and autoimmune diseases. This effort opens potential for strategic partnerships. It is a time-consuming, capital intensive effort, to transform an entity into a provider of platform, products and services in a relatively short time. We believe when the transition is completed over the next year that the Company will look very different than it does today, with an anticipated return to revenue and margin growth.”
Fiscal 2018 Operating Results
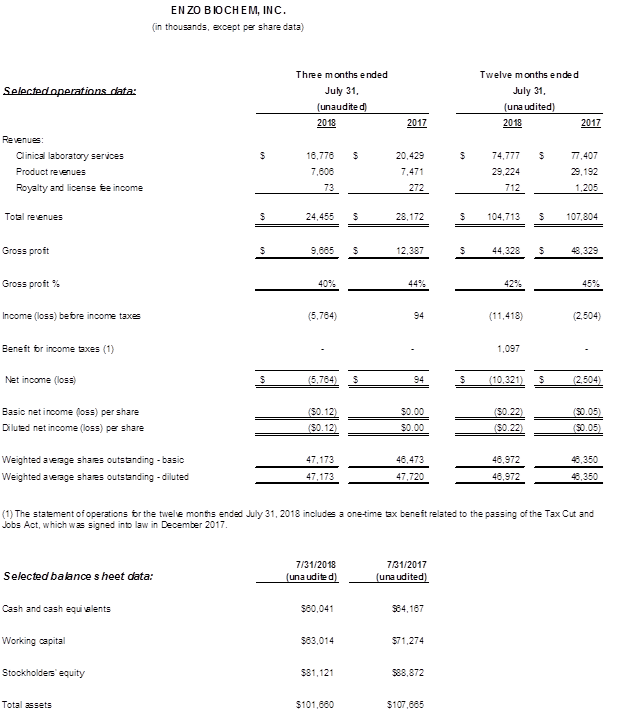
- Total revenues were $104.7 million, compared to $107.8 million in the prior year, a decrease of $3.1 million, or 3%. Clinical services revenues were $74.8 million, compared to $77.4 million in the prior year, a decrease of $2.6 million or 3% due to lower insurance reimbursement payments and shifts in test mix to lower esoteric testing verses high genetic testing in the prior year. Total diagnostic testing volume, measured by the number of accessions reported, increased 4% year over year. Clinical products and royalties revenue was $29.9 million compared to $30.4 million in the prior year. The decline year over year resulted from lower product royalties from an agreement that expired in April 2018.
- Consolidated gross margins were 42% compared with 45% in the prior year. Clinical services gross margins were 39% compared to 41% a year ago. Gross margins in the current year were negatively impacted by lower revenue from Clinical Services as noted above. Clinical products and royalties gross margin was 52% compared to 54% in the prior year period.
- Operating expenses totaled $56.5 million, up 10% compared to $51.4 million a year ago. The increase reflected legal fee expenses in anticipation of a patent infringement and contract related trial, where Enzo is plaintiff that may occur next calendar year. Total legal expenses were $5.1 million compared to $1.7 million in the prior year. Selling and general administrative expenses (SG&A) as well as research and development (R&D) expenses were slightly higher year over year in support of the Company’s growth strategies. As a percentage of revenue, SG&A was 42% compared to 41% in the prior year and R&D expenses were flat year over year.
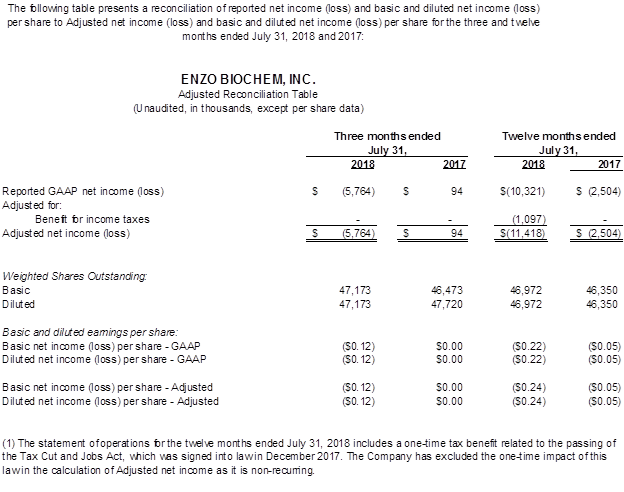
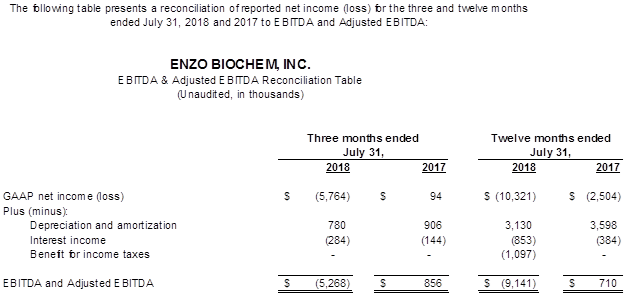
- The GAAP and Non-GAAP net loss was $10.3 million and $11.4 million, respectively, compared to $2.5 million a year ago. The GAAP net loss per share was $0.22, compared to $0.05 a year ago, and the Non-GAAP loss per share was $0.24. There were no Non-GAAP adjustments in the prior year. EBITDA was a loss of $9.1 million compared to earnings of $0.7 million a year ago.
Fourth Quarter Operating Results
- Total revenues were $24.5 million, compared to $28.2 million in the prior year, a decrease of $3.7 million. Clinical services revenues were $16.8 million, compared to $20.4 million in the prior year, a decrease of $3.7 million. Approximately $1.7 million of the current quarter’s revenue decline relates to a previously reported account loss with the remaining decline due to lower insurance reimbursement payments and shifts in test mix away from high valued genetic testing in the prior year. Total diagnostic testing volume, measured by the number of accessions reported, decreased 3% year over year, however it increased 3% sequentially compared to the third fiscal quarter of 2018. Clinical products and royalties revenue was approximately $7.7 million in both the current year and prior year periods.
- Consolidated gross margins were 40% compared with 44% in the prior year. Clinical services gross margins were 33% compared to 41% a year ago. Gross margins in the current year were negatively impacted by lower revenue from Clinical Services as noted above. Clinical products and royalties gross margins increased 120 basis points year over year to 54%.
- Operating expenses totaled $15.4 million, up 19% compared to $12.9 million from a year ago. The increase reflected higher legal fee expenses of $0.9 million in anticipation of the aforementioned patent infringement and contract related trial, increased allowances for bad debts of $0.9 million and higher SG&A expenses of $0.6 million. R&D expenses were flat year over year.
- The GAAP and Non-GAAP net loss was $5.8 million compared to breakeven a year ago. The GAAP and Non-GAAP net loss per share was $0.12 and compared to $0.05 a year ago. There were no Non-GAAP adjustments in the current and prior year. EBITDA was a loss of $5.3 million compared to EBITDA earnings of $0.9 million a year ago.
Conference Call
The Company will conduct a conference call Tuesday, October 16, 2018 at 8:30 AM ET. The call can be accessed by dialing 1-888-459-5609. International callers can dial 1-973-321-1024. Please reference PIN number 6096026.
Interested parties may also listen over the Internet at: https://tinyurl.com/y8kzakd2
To listen to the live call, individuals should go to the website at least 15 minutes early to register, download and install any necessary audio software. Any pop up blocker installed on your PC should be disabled while accessing the webcast. A rebroadcast of the call will be available starting approximately two hours after the conference call ends, through 12 AM (E.T.) Tuesday, October 30, 2018. The replay of the conference call can be accessed by dialing 1-855-859-2056 (International callers can dial 1-404-537-3406) and, when prompted, use the same PIN number 6096026.
Adjusted Financial Measures
To comply with Regulation G promulgated pursuant to the Sarbanes-Oxley Act, Enzo Biochem attached to this news release and will post to the Company's investor relations web site (www.enzo.com) any reconciliation of differences between GAAP and Adjusted financial information that may be required in connection with issuing the Company's quarterly financial results.
The Company uses EBITDA as a measure of performance to demonstrate earnings exclusive of interest, taxes, depreciation and amortization. Adjustments to EBITDA are for items of a non-recurring nature and are reconciled on the table provided. The Company manages its business based on its operating cash flows. The Company, in its daily management of its business affairs and analysis of its monthly, quarterly and annual performance, makes its decisions based on cash flows, not on the amortization of assets obtained through historical activities. The Company, in managing its current and future affairs, cannot affect the amortization of the intangible assets to any material degree, and therefore uses EBITDA as its primary management guide. Since an outside investor may base its evaluation of the Company's performance based on the Company's net loss not its cash flows, there is a limitation to the EBITDA measurement. EBITDA is not, and should not be considered, an alternative to net loss, loss from operations, or any other measure for determining operating performance of liquidity, as determined under accounting principles generally accepted in the United States (GAAP). The most directly comparable GAAP reference in the Company's case is the removal of interest, taxes, depreciation and amortization.
We refer you to the tables attached to this press release which includes reconciliation tables of GAAP to Adjusted net income (loss) and EBITDA to Adjusted EBITDA.
About Enzo Biochem
Enzo Biochem is a pioneer in molecular diagnostics, leading the convergence of clinical laboratories, life sciences and intellectual property through the development of unique diagnostic platform technologies that provide numerous advantages over previous standards. A global company, Enzo Biochem utilizes cross-functional teams to develop and deploy products, systems and services that meet the ever-changing and rapidly growing needs of health care today and into the future. Underpinning Enzo Biochem’s products and technologies is a broad and deep intellectual property portfolio, with patent coverage across a number of key enabling technologies.
Except for historical information, the matters discussed in this news release may be considered "forward-looking" statements within the meaning of Section 27A of the Securities Act of 1933, as amended and Section 21E of the Securities Exchange Act of 1934, as amended. Such statements include declarations regarding the intent, belief or current expectations of the Company and its management, including those related to cash flow, gross margins, revenues, and expenses which are dependent on a number of factors outside of the control of the Company including, inter alia, the markets for the Company’s products and services, costs of goods and services, other expenses, government regulations, litigation, and general business conditions. See Risk Factors in the Company’s Form 10-K for the fiscal year ended July 31, 2018. Investors are cautioned that any such forward-looking statements are not guarantees of future performance and involve a number of risks and uncertainties that could materially affect actual results. The Company disclaims any obligations to update any forward-looking statement as a result of developments occurring after the date of this press release.
###
Contact:
For: Enzo Biochem, Inc.
Steve Anreder, 212-532-3232 or Michael Wachs, CEOcast, Inc., 212-732-4300
steven.anreder@anreder.com mwachs@ceocast.com