NEW YORK, NY, March 12, 2018 – Enzo Biochem Inc. (NYSE:ENZ) today announced results for the fiscal quarter and six months ended January 31, 2018.
Quarter Highlights (Year over Year)
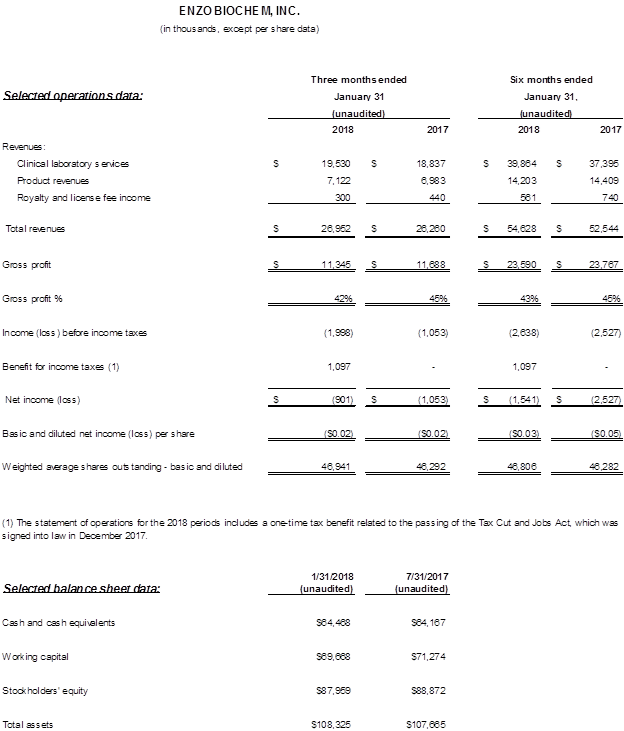
- Total revenues for the second fiscal quarter increased to $27.0 million, or 3%, from $26.3 million in the prior year period.
- Clinical Lab revenues for the quarter totaled $19.5 million, an increase of 4% over the prior year period, despite severe weather impacting operations. Enzo Life Sciences revenue was $7.4 million, unchanged over the year ago.
- Gross margins were 42% in the current year quarter, lower than 44% in the prior year period due to adverse weather noted above and lower genomic product sales.
- Heavier than usual increased legal expenses of $1.7 million resulted in an operating loss of ($3.1) million compared to ($1.0) million a year ago.
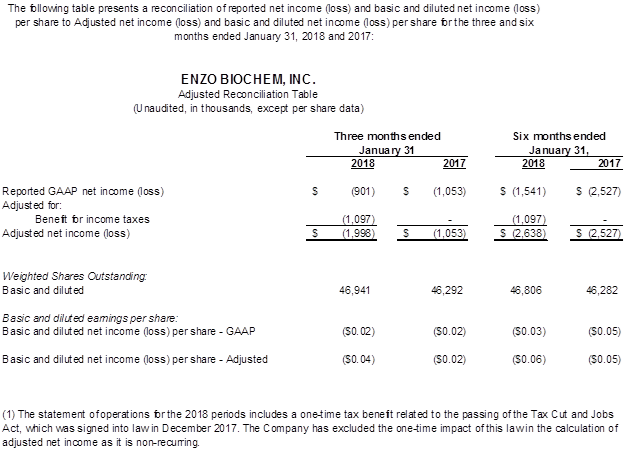
- GAAP net loss declined to ($0.9) million or ($0.02) per share, compared to ($1.0) million or ($0.02) per share a year ago, and for six months amounted to ($1.6) million or ($0.03) per share, compared to ($2.5) million or ($0.05) per share, a $0.9 million improvement.
- Total cash and cash equivalents at January 31, 2018 was $64.5 million, an increase of $0.3 million from July 31, 2017, and consolidated cash flow from operations was $0.8 million for the six months ended January 31, 2018, an increase of $3.7 million over the prior year period. Working capital stands at nearly $70 million as of January 31, 2018.
- During the quarter, the Company fully integrated and validated its AmpiProbe® platform for use in its laboratory which is expected to result in significant savings in the cost of laboratory services in future periods.
- Interest in Enzo’s now completed 13-analyte women’s diagnostics health panel continues to mount, reinforcing build-up of physician servicing network in northeast service territory.
- In addition to continued emphasis on product development, the quarter’s activity was focused on increased marketing, including additions to and training of our sales staff, and appearances at major industry conferences and meetings to promote the Company’s highly efficient, higher margin molecular diagnostic test alternatives to profit-squeezed independent labs.
Barry Weiner, President, Comments
“The second fiscal quarter of 2018 has been an especially productive period. While we continued to gain ground with our AmpiProbe® products and other New York State Department of Health approved diagnostics designed to allay cost pressures affecting the nation’s independent clinical laboratories, severe winter weather cut into our clinical laboratory operations revenues. In addition, in anticipation of a trial in New York’s Federal Southern District in litigation with Roche, following a favorable Markman patent-related decision by the court for Enzo, stepped-up depositions involving extensive expert testimony resulted in a substantial increase in litigation expenses. This case involves both patent and contract issues.
“Total revenues continue to grow, with Clinical Labs quarterly results improving year over year despite the harsh weather that affected operations of approximately two days resulting in lost revenues. Product revenues at Life Sciences also advanced, underscoring the benefits starting to derive from our strategic program to increase both services and product revenues. Our financial position and balance sheet remain strong providing the necessary capital from positive cash flow to grow organically and make long term capital investments.
“The expected new reduced PAMA Medicare reimbursements took effect in January further enforcing our strategy in helping laboratories to improve their margin utilizing our program of low cost products and services. Our focus remains on serving independent labs as an integrated, growth-oriented molecular diagnostics company, and a low-cost medically related assay provider and reference service organization. Put simply, with our current approved platforms and assays and those in our pipeline, we expect to be a lead supplier of affordable and reliable diagnostic testing either by product or service. Towards that end, we are diligently expanding and investing in our marketing program directed at independent labs and hospitals. The superior effectiveness and utility of our products that are compatible with existing in-house diagnostic systems has been demonstrated in various studies. We will adhere to those high standards of sensitivity and economy as we build our comprehensive testing line-up moving forward.”
Second Quarter Results
The quarter’s total revenues amounted to approximately $27 million, compared to $26.3 million a year ago, an increase of 3%. Gross profit was $11.3 million or 42% of total revenue. Total operating expenses were $14.4 million and included selling and general and administrative expenses of $11.1 million, or 41% of revenue.
As noted, a favorable Markman decision in our New York litigation with Roche resulted in a significant increase in litigation expenditures to $1.7 million, compared with $370,000 a year ago. After slightly higher interest income, a foreign currency gain, and a $1.1 million tax benefit attributable to the Tax Cuts and Jobs Act of 2017, GAAP net loss amounted to $(0.9) million, or $(0.02) per share, compared to the year ago GAAP net loss of $(1.1) million or $(0.02) per share. Adjusted net loss amounted to $(2.0) million or $(0.04) per share compared to Adjusted net loss of $(1.1) million or $(0.02) per share a year ago. EBITDA (earnings before interest, taxes, depreciation and amortization) and Adjusted EBITDA were both a loss of ($1.4) million, compared to ($0.2) million in the prior year period due largely to increased legal expenses.
As of January 31, 2018, cash and cash equivalents were $64.5 million with working capital of nearly $70 million. Cash flow from operations for the six months was $0.8 million, an increase of $3.7 million over the prior year period.
First Half Results
Total revenues were $54.6 million or 4% higher than prior year. Gross profit totaled $23.6 million, compared to $23.8 million a year ago, with gross margins of 43% and 45%, respectively. SG&A of $22.0 million increased $0.7 million, but as a percentage of sales declined to 40% from 43% a year ago. With legal expenses increasing to $2.1 million, from $0.7 million a year ago, the operating loss amounted to ($3.7) million, compared with ($2.2) million. The six month GAAP net loss totaled ($1.5) million, or ($0.03) per share, down from ($2.5) million, or ($0.05) per share, a 38% improvement.
Segment Results - Quarter
Enzo Clinical Labs revenues were $19.5 million, an increase of 4%, from $18.8 million in the prior year. The increase in revenue was offset in part by adverse winter weather and a severe Flu season in the Northeast that essentially curtailed patient visits to physicians and clinics by approximately two days during the quarter, consequently reducing the number of tests submitted and processed. As a result, gross margins were 40%, compared to 41% in the prior year period. SG&A increased approximately $210,000 due to headcount and costs in client and billing services, but as a percentage of revenues remained at 31%. Operating income was $0.9 million, versus $1.2 million in the prior year period. Enzo Life Sciences revenues were $7.4 million, unchanged over the year ago.
Gross profit equaled 48% compared to 53% a year ago due to lower genomic product sales and a decline in royalty income. SG&A remained flat at approximately $2.9 million, though as a percentage of revenues declined 10 basis points to 39%. Operating income amounted to $51,000, versus $381,000 a year ago due to lower margins on product sales.
Conference Call
The Company will conduct a conference call Tuesday, March 13, 2018 at 8:30 AM ET. The call can be accessed by dialing 1-888-459-5609. International callers can dial 1-973-321-1024. Please reference PIN number 9386236.
Interested parties may also listen over the Internet at: https://tinyurl.com/yb9wet9f
To listen to the live call on the Internet, please go to the web site at least fifteen minutes early to register, download and install any necessary audio software. For those who cannot listen to the live broadcast, a replay will be available approximately two hours after the end of the live call, through midnight (ET) on March 27, 2018. The replay of the conference call can be accessed by dialing 1-855-859-2056, and when prompted, use PIN number 9386236. International callers can dial 1-404-537-3406, using the same PIN number.
ADJUSTED Financial Measures
To comply with Regulation G promulgated pursuant to the Sarbanes-Oxley Act, Enzo Biochem attached to this news release and will post to the Company's investor relations web site (www.enzo.com) any reconciliation of differences between GAAP and Adjusted financial information that may be required in connection with issuing the Company's quarterly financial results.
The Company uses EBITDA as a measure of performance to demonstrate earnings exclusive of interest, taxes, depreciation and amortization. Adjustments to EBITDA are for items of a non-recurring nature and are reconciled on the table provided. The Company manages its business based on its operating cash flows. The Company, in its daily management of its business affairs and analysis of its monthly, quarterly and annual performance, makes its decisions based on cash flows, not on the amortization of assets obtained through historical activities. The Company, in managing its current and future affairs, cannot affect the amortization of the intangible assets to any material degree, and therefore uses EBITDA as its primary management guide. Since an outside investor may base its evaluation of the Company's performance based on the Company's net loss not its cash flows, there is a limitation to the EBITDA measurement. EBITDA is not, and should not be considered, an alternative to net loss, loss from operations, or any other measure for determining operating performance of liquidity, as determined under accounting principles generally accepted in the United States (GAAP). The most directly comparable GAAP reference in the Company's case is the removal of interest, taxes, depreciation and amortization.
We refer you to the tables attached to this press release which includes reconciliation tables of GAAP to Adjusted net income (loss) and EBITDA to Adjusted EBITDA.
About Enzo Biochem
Enzo Biochem is a pioneer in molecular diagnostics, leading the convergence of clinical laboratories, life sciences and intellectual property through the development of unique diagnostic platform technologies that provide numerous advantages over previous standards. A global company, Enzo Biochem utilizes cross-functional teams to develop and deploy products, systems and services that meet the ever-changing and rapidly growing needs of health care today and into the future. Underpinning Enzo Biochem’s products and technologies is a broad and deep intellectual property portfolio, with patent coverage across a number of key enabling technologies.
Except for historical information, the matters discussed in this news release may be considered "forward-looking" statements within the meaning of Section 27A of the Securities Act of 1933, as amended and Section 21E of the Securities Exchange Act of 1934, as amended. Such statements include declarations regarding the intent, belief or current expectations of the Company and its management, including those related to cash flow, gross margins, revenues, and expenses which are dependent on a number of factors outside of the control of the Company including, inter alia, the markets for the Company’s products and services, costs of goods and services, other expenses, government regulations, litigation, and general business conditions. See Risk Factors in the Company’s Form 10-K for the fiscal year ended July 31, 2017. Investors are cautioned that any such forward-looking statements are not guarantees of future performance and involve a number of risks and uncertainties that could materially affect actual results. The Company disclaims any obligations to update any forward-looking statement as a result of developments occurring after the date of this press release.
###
Contact:
For: Enzo Biochem, Inc.
Steve Anreder, 212-532-3232 or Michael Wachs, CEOcast, Inc., 212-732-4300
steven.anreder@anreder.com mwachs@ceocast.com